Abstract
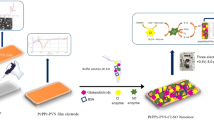
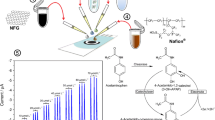
The aim of this work was to create the new biosensor of alanine transaminase (ALT) activity detection in water samples. A platinum disk electrode was used to transduce chemical reactions into amperometry measured signal. Nanosize polyphenylendiamine (PPD) was applied to improve the selectivity of the transducer. In the work, the most effective isomer PD was selected and the stability of the additional membrane based on it was checked. The bioselective element was formed by the immobilization of glutamate oxidase on the surface of the transducer covered by a semipermeable P-meta-PD membrane. There was reported the method of ALT activity measurement. There was also reported the optimal conditions to create this biosensor (including enzyme immobilization parameters and substrates concentrations). The analytical characteristics were investigated including the sensitivity, the linear range, the minimum limit of detection, time of measurement, duration of analysis, stability, etc. Also, there was explored the biosensor selectivity to different interferents, e.g., electroactive molecules and amino acids. For the purpose to test the fundamental possibility of this biosensor to measure the ALT activity in real samples, the operation of the biosensor was tested in a solution with an ionic composition close to serum.












Similar content being viewed by others
Data Availability
Raw data and derived data supporting the findings of this study are available from the corresponding author Mruga D. on request.
References
An L (2015) Detection of glutamate, glutamine, and glutathione by radiofrequency suppression and echo time optimization at 7 tesla. Magn Reson Med 73(2):451–458. https://doi.org/10.1002/mrm.25150
Bailey W, DeRouen T, Ziskind M et al (1975) Autoanalytic (Colormetric) determinations of SGOT in isoniazid recipients are reliable. Am Rev Respir Dis 111(2):237–238
Bonizzoni M, Fabbrizzi L, Piovani G et al (2004) Fluorescent detection of glutamate with a dicopper(II) polyamine cage. Tetrahedron 60(49):11159–11162. https://doi.org/10.1016/j.tet.2004.08.102
Catz S, Carreras M, Poderoso J (1995) Nitric oxide synthase inhibitors decrease human polymorphonuclear leukocyte luminol-dependent chemiluminescence. Free Radic Biol Med 19(6):741–748. https://doi.org/10.1016/0891-5849(95)00065-6
Chang K, Hsu W, Chen H et al (2003) Determination of glutamate pyruvate transaminase activity in clinical specimens using a biosensor composed of immobilized L-glutamate oxidase in a photo-crosslinkable polymer membrane on a palladium-deposited screen-printed carbon electrode. Anal Chim Acta 481(2):199–208. https://doi.org/10.1016/S0003-2670(03)00093-X
Chenu C (2002) Glutamatergic regulation of bone remodeling. J Musculoskelet Neuronal Interact 2(3):282–284 (PMID: 15758455)
Duran B, Bereket G, Duran M (2012) Electrochemical synthesis and characterization of poly(m-phenylenediamine) films on copper for corrosion protection. Prog Org Coat 73(1–2):162–168. https://doi.org/10.1016/j.porgcoat.2011.10.008
Ferguson D (1988) Physiology for dental student. Butterworth-Heinemann, Blood plasma, 42–53. https://doi.org/10.1016/B978-0-7236-0725-0.50006-X
Giannini EG, Testa R, Savarino V (2005) Liver enzyme alteration: a guide for clinicians. CMAJ 172(3):367–379. https://doi.org/10.1503/cmaj.1040752
Stepulak A, Rola R, Polberg K et al (2014) Glutamate and its receptors in cancer. J Neural Transm (Vienna) 121(8):933–944. https://doi.org/10.1007/s00702-014-1182-6
Han M, Kopec W, Solovyov I et al (2017) Glutamate water gates in the ion binding pocket of Na+ Bound Na+, K+-ATPase. Sci Rep 7:39829. https://doi.org/10.1038/srep39829
Janasek D, Spohn U (1999) Chemiluminometric flow injection analysis procedures for the enzymatic determination of L-alanine, α-ketoglutarate and L-glutamate. Biosens Bioelectron 14(2):123–129. https://doi.org/10.1016/S0956-5663(98)00115-8
Jin L, Li D, Alesi N et al (2015) Glutamate dehydrogenase 1 signals through antioxidant glutathione peroxidase 1 to regulate redox homeostasis and tumor growth. Cancer Cell 27(2):257–270. https://doi.org/10.1016/j.ccell.2014.12.006
Kasraeian M, Asadi N, Vafaei H et al (2018) Evaluation of serum biomarkers for detection of preeclampsia severity in pregnant women. Pak J Med Sci 34(4):869–873
Khampha W, Yakovleva J, Isarangkul D et al (2004) Specific detection of L-glutamate in food using flow-injection analysis and enzymatic recycling of substrate. Anal Chim Acta 518(1–2):127–135. https://doi.org/10.1016/j.aca.2004.05.048
Kim HJ, Kim SY, Shin SP et al (2020) Immunological measurement of aspartate/alanine aminotransferase in predicting liver fibrosis and inflammation. Korean J Intern Med 35(2):320–330. https://doi.org/10.3904/kjim.2018.214
Kondrat R (2002) In vivo microdialysis and gas-chromatography/mass-spectrometry for 13C-enrichment measurement of extracellular glutamate in rat brain. J Neurosci Methods 120(2):179–192. https://doi.org/10.1016/s0165-0270(02)00201-7
Kucherenko I, Soldatkin O, Kasap B et al (2012) Elaboration of urease adsorption on silicalite for biosensor creation. Electroanalysis 24(6):1380–1385. https://doi.org/10.1002/elan.201200056
Kucherenko I, Soldatkin O, Kucherenko D et al (2019) Advances in nanomaterial application in enzyme-based electrochemical biosensors: a review. Nanoscale Adv 1(12):4560–4577. https://doi.org/10.1039/c9na00491b
Kwan R, Hon P, Mak K et al (2004) Amperometric determination of lactate with novel trienzyme/poly(carbamoyl) sulfonate hydrogel-based sensor. Biosens Bioelectron 19(12):1745–1752. https://doi.org/10.1016/j.bios.2004.01.008
Lehmann-Werman R, Magenheim J, Moss J et al (2018) Monitoring liver damage using hepatocyte-specific methylation markers in cell-free circulating DNA. JCI Insight. https://doi.org/10.1172/jci.insight.120687
Lehninger A, Nelson D, Cox M (1993) Principles of biochemistry. 2nd Ed 1993 Amino Acid Oxid Product Urea. https://doi.org/10.1002/elan.202060449
Lehninger A, Nelson D, Cox M (1982) Principles of biochemistry, 1st edition. Worth Publishers, New York, 175. https://doi.org/10.1016/0307-4412(83)90020-1
Li L, Wang M, Chen S et al (2016) A urinary metabonomics analysis of long-term effect of acetochlor exposure on rats by ultra-performance liquid chromatography/mass spectrometry. Pestic Biochem Phys 128:82–88. https://doi.org/10.1016/j.pestbp.2015.09.013
Li J, Yuan Z, Wang Q et al (2019) Meta-analysis of overall incidence and risk of ALK inhibitors-induced liver toxicities in advanced non-small-cell lung cancer. Medicine (baltimore) 98(1):13726. https://doi.org/10.1097/MD.0000000000013726
Lippi U, Guidi G (1970) A new colorimetric ultramicromethod for serum glutamic-oxalacetic and glutamic-pyruvic transaminase determination. Clin Chim Acta 28(3):431–437. https://doi.org/10.1016/0009-8981(70)90069-0
Liu X, Mei X, Yang J et al (2022) Hydrogel-involved colorimetric platforms based on layered double oxide nanozymes for point-of-care detection of liver-related biomarkers. ACS Appl Mater Interfaces 14(5):6985–6993. https://doi.org/10.1021/acsami.1c21578
Mahale A, Prabhu S, Nachiappan M et al (2018) Clinical relevance of reporting fatty liver on ultrasound in asymptomatic patients during routine health checkups. J Int Med Res 46(11):4447–4454. https://doi.org/10.1177/0300060518793039
Marquette C, Degiuli A, Blum L (2003) Electrochemiluminescent biosensors array for the concomitant detection of choline, glucose, glutamate, lactate, lysine and urate. Biosens Bioelectron 19(5):433–439. https://doi.org/10.1016/s0956-5663(03)00225-2
Mattson M (2018) Glutamate and neurotrophic factors in neuronal PlACTicity and disease. Ann NY Acad Sci 144:97–112. https://doi.org/10.1196/annals.1418.005
Mayer M, Baeumner AJ (2019) A megatrend challenging analytical chemistry: biosensor and chemosensor concepts ready for the internet of things. Chem Rev 119(13):7996–8027. https://doi.org/10.1021/acs.chemrev.8b00719
Mayer D, Spielman D (2005) Detection of glutamate in the human brain at 3 T using optimized constant time point resolved spectroscopy. Magn Reson Med 54:2. https://doi.org/10.1002/mrm.20571
Mousa HM, Aggas JR, Guiseppi-Elie A (2019) Electropolymerization of aniline and (N-phenyl-o-phenylenediamine) for glucose biosensor application. Mater Lett 238:267–270. https://doi.org/10.1016/j.matlet.2018.12.012
Mruga D, Soldatkin O, Paliienko K et al (2021) Optimization of the design and operating conditions of an amperometric biosensor for glutamate concentration measurements in the blood plasma. Electroanalysis 33(5):1299–1307. https://doi.org/10.1002/elan.202060449
Okumoto S, Looger L, Micheva R et al (2005) Detection of glutamate release from neurons by genetically encoded surface-displayed FRET nanosensors. Proc Natl Acad Sci USA 102(24):8740–8745. https://doi.org/10.1073/pnas.0503274102
Oldenziel W, Westerink B (2005) Improving glutamate microsensors by optimizing the composition of the redox hydrogel. Anal Chem 77(17):5520–5528. https://doi.org/10.1021/ac0580013
Perales M, Sener A, Malaisse W (1992) Radioisotopic Assay of Aspartate and Alanine Aminotransferase. Clin Biochem 25(2):105–107. https://doi.org/10.1016/0009-9120(92)80052-I
Peshkova V, Kucherenko I, Soldatkin O et al (2013) Method of testing and optimization of amperometric transducers. Sens Electr Microsyst Technol 10:3
Pyeshkova VM, Dudchenko OY, Soldatkin OO et al (2022) Development of three-enzyme lactose amperometric biosensor modified by nanosized poly (meta-phenylenediamine) film. Appl Nanosci 12:1267–1274. https://doi.org/10.1007/s13204-021-01859-8
Rahman A, Kwon N, Won M et al (2005) Functionalized conducting polymer as an enzyme-immobilizing substrate: an amperometric glutamate microbiosensor for in vivo measurements. Anal Chem 7(15):4854–4860. https://doi.org/10.1021/ac050558v
Raya AG, Zuniga RC, Salodo EM et al (2019) Isolated elevation of aspartate aminotransferase (AST) in an asymptomatic patient due to macro-AST. JCLA 33:22690. https://doi.org/10.1002/jcla.22690
Lala V, Zubair M, Minter DA (2022) Liver Function Tests. StatPearls. https://www.ncbi.nlm.nih.gov/books/NBK482489/
The University of California San Francisco (2019) Plasma amino acids. UCSFHealth, internet resource https://www.ucsfhealth.org/medical-tests/plasma-amino-acids
Shin H (2017) Metabolic profiling of tyrosine, tryptophan, and glutamate in human urine using gas chromatography-tandem mass spectrometry combined with single SPE cleanup. J Chromatogr B Analyt Technol Biomed Life Sci 1051:97–107. https://doi.org/10.1016/j.jchromb.2017.03.015
Soldatkin O, Soldatkina O, Piliponskiy I et al (2021) Application of gold nanoparticles for improvement of analytical characteristics of conductometric enzyme biosensors. Appl Nanosci 12:995–1003. https://doi.org/10.1007/s13204-021-01807-6
Soldatkina O, Kucherenko I, Soldatkin O et al (2013) Development of electrochemical biosensors with various types of zeolites. Appl Nanosci (switzerland) 9(5):737–747. https://doi.org/10.1007/s13204-018-0725-9
Soldatkina O, Kucherenko I, Pyeshkova V et al (2017) Improvement of amperometric transducer selectivity using nanosized phenylenediamine films. Nanoscale Res Lett 12(1):594. https://doi.org/10.1186/s11671-017-2353-9
Son J, Lyssiotis S, Ying H et al (2013) Glutamine supports pancreatic cancer growth through a Kras-regulated metabolic pathway. Nature 496(7443):101–105. https://doi.org/10.1038/nature12040
Suman S, Singhal R, Sharma A et al (2004) Development of a lactate biosensor based on conducting copolymer bound lactate oxidase. Sens Actu B Chem 107(2):768–772. https://doi.org/10.1016/j.snb.2004.12.016
Tan D, Xiang W, Wang H et al (2022) Optical assay using B-doped core–shell Fe@BC nanozyme for determination of alanine aminotransferase. Microchim Acta 189:147. https://doi.org/10.1007/s00604-021-05056-w
Tao J-Z, Xu G-R, Hao H-L et al (2013) Poly(m-phenylenediamine)–Prussian blue hybrid film formed by one-step electrochemical deposition for glucose biosensor. J Electroanal Chem 689:96–102. https://doi.org/10.1016/j.jelechem.2012.09.043
Teng J, Wei Y, Su F et al (2015) Evaluating of physiological chemical levels in blood to assess the risk of morbidity and mortality of ischemic cardiovascular disease. Int J Environ Res Public Health 12(9):11549–11559. https://doi.org/10.3390/ijerph120911549
Toney MD (2013) Encyclopedia of biological chemistry II. Elsevier, Pyridoxal Phosphate, 708–11. https://doi.org/10.1016/B978-0-12-378630-2.00024-4
Trister A, Scott J, Rockne R et al (2014) Response to Tumor cells in search for glutamate: an alternative explanation for increased invasiveness of IDH1 mutant gliomas. Neuro Oncol 16(12):1670–1671. https://doi.org/10.1093/neuonc/nou290
van Lith S, Molenaar R, Noorden C et al (2014) Tumor cells in search for glutamate: an alternative explanation for increased invasiveness of IDH1 mutant gliomas. Neuro Oncol 16(12):1669–1670. https://doi.org/10.1093/neuonc/nou152
Wang HL, Chu CH, Tsai SJ et al (2016) Aspartate aminotransferase and alanine aminotransferase detection on paper-based analytical devices with inkjet printer-sprayed reagents. Micromachines 7(1):7. https://doi.org/10.3390/mi7010009
Xuan G, Oh S, Choi E (2003) Development of an electrochemical immunosensor for alanine aminotransferase. Biosens Bioelectron 19(4):365–371. https://doi.org/10.1016/s0956-5663(03)00194-5
Yang J, He X, Huang B et al (2018) Rule of changes in serum GGT levels and GGT/ALT and AST/ALT ratios in primary hepatic carcinoma patients with different AFP levels. Cancer Biomark 21(4):743–746. https://doi.org/10.3233/CBM-170088
Severin 2002 with The University of California San Francisco (2019) Plasma amino acids. UCSFHealth, internet resource https://www.ucsfhealth.org/medical-tests/plasma-amino-acids
Severin 2014 with Lala V, Zubair M, Minter DA (2022) Liver Function Tests. StatPearls. https://www.ncbi.nlm.nih.gov/books/NBK482489/
Acknowledgements
The work was carried out at the expense of a grant of the National Academy of Sciences of Ukraine to research laboratories of young scientists of the NASU for conducting research in the priority directions of the development of science and technology in 2022–2023.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical statements
No animal experiments or human participants were involved in that study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mruga, D., Dzyadevych, S. & Soldatkin, O. High-selective alanine transaminase-sensitive biosensor based on nanosize semipermeable poly-meta-phenylenediamine membrane. Appl Nanosci 13, 6939–6949 (2023). https://doi.org/10.1007/s13204-023-02810-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-023-02810-9